Ever-growing momentum
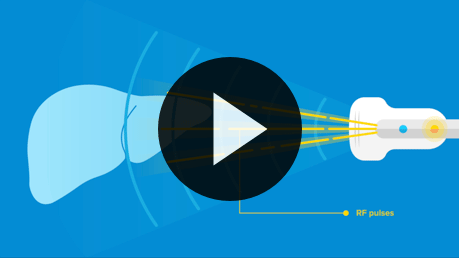
The photo-acoustic ultrasound technology we first created for laboratory research of complex diseases—Nexus 128—led us to an even more powerful enhancement for clinical use: Thermo-Acoustic Enhanced UltraSound (TAEUS®).
TAEUS®
TAEUS® enables clinicians to see tissue in ways previously possible only with CT or MRI—at a fraction of the cost, and at the point of care.
Explore TAEUS® >
Liver Disease
Non-Alcoholic Fatty Liver Disease (NAFLD) is difficult to identify and monitor. ENDRA is enhancing ultrasound to change that.
Explore liver applications >Who we are
We are a passionate group of engineers, healthcare professionals and commercial leaders with decades of experience in solving complex technical problems. We’re working to uncover cost-effective solutions to some of the world’s most difficult healthcare challenges.
Endra Explained
Meet Mary
Learn how ENDRA is using ultrasound to help make difficult-to-diagnose diseases easier to find and treat.
Watch the video >What is TAEUS®?
Thermo-Acoustic Enhanced UltraSound will revolutionize what doctors can see and do at the patient bedside.
Watch the video >Our partners
Our partnerships with leading medical researchers, engineers and manufacturers support ENDRA’s commitment to bringing TAEUS® to market. We benefit from each of these companies’ deep expertise.
Clinical partners

Rocky Vista University
Parker, CO
UPMC
Pittsburgh, PA
Centre Hospitalier Universitaire d'Angers
Angers, France
Johannes Gutenberg University
Frankfort, Germany
Inselspital University Hospital
Bern, Switzerland
Medical College of Wisconsin
Wauwatosa, WIIndustry and research partners

Hepion Pharmaceuticals
Edison, NJ
GE
Chicago, IL
Starfish
Victoria, B.C.
Cimtec
London, Ont.“We believe that ENDRA’s technology has the potential to bring significant new capabilities to ultrasound—which aligns well with GE Healthcare’s mission of increasing access to high-quality, cost-effective healthcare”